Often there is confusion between the term nutrition and the term nutraceuticals. They often tend to be mistaken for each other when it comes to communicating information by companies seeking to improve the positioning of their fruit and vegetable and processed products.
 Tiziana Pecora
Tiziana Pecora
Another source of confusion is the existing legislative framework, which must be interpreted on the basis of existing Italian and European laws. For this reason, we interviewed Tiziana Pecora, a specialist in Pharmacology and an expert in regulatory disciplines, who pointed out that "legislative intervention is needed, especially at a EU level, in order to fill the regulatory gaps and uncertainties, thus adapting the regulatory framework to the new market and consumer needs and making it possible to sell and advertise these products correctly."
Health and nutrition are strongly linked, and nutraceuticals play a key role. The definition of a nutraceutical was provided by Stephen L. DeFelice in 1989, as food (or part of food) capable of providing medical or health benefits, including the prevention and/or treatment of disease.
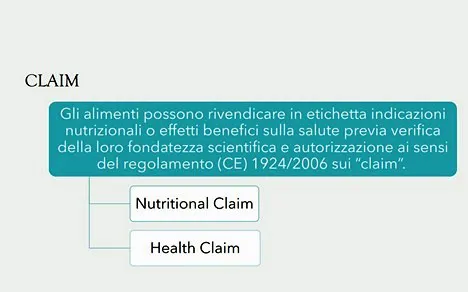
"Although the term nutraceutical is not acknowledged by the EU legislation, it refers to a food which is able to claim beneficial effects on the body thanks to the content of particular constituents. In business, we refer to nutraceuticals as functional foods and food supplements. They are considered to be foodstuffs and, as such, are regulated by the food law, i.e., as part of the legal regulations governing the production, communication and trade of foodstuffs,” continued Pecora.

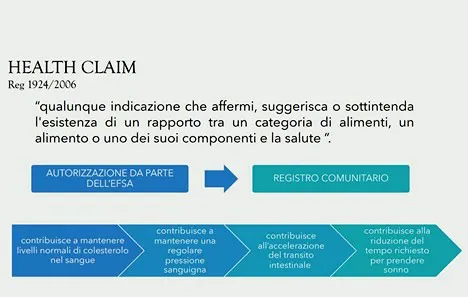
Even functional food is not defined by European legislation and it is described only in a Consensus Document of the European Commission in agreement with Fufose, Functional Food Science in Europe as a substance that possesses additional effects due to the presence of components, generally non-nutritious, that interact selectively with one or more physiological functions of the body, in such a way that an improvement in the state of health and well-being and/or a reduction in the risk of disease are evident", explained our interviewee.
"As defined by the Legislative Decree 169/2004, food supplements are food products intended to supplement the common diet and therefore constitute a concentrated source of nutrients, such as vitamins and minerals, or of other substances with a nutritional or physiological effect, in particular, but not exclusively, amino acids, essential fatty acids, fibre and extracts of vegetable origin, both single and multi-compound, in pre-dosed forms, i.e. the classic pharmaceutical forms for oral use (capsules, tablets, powders, etc.), intended to be used by the consumer for the intake of nutrients,” added Tiziana Pecora.
While some foods have an informational advertising on the health benefits, that is, they can indicate a substance that makes them functional, other foods don't. In that case, communication is based on aspects that are regulated. In the case of other fruit and vegetable products (e.g., tomatoes), whose substances do not have a statement as required by European legislation, the undoubted health benefits cannot be denied. The key lies in correct communication. While the beneficial combination of some products (e.g., citrus fruit - vitamin C) is well established, for others (e.g., brassicas, apples, etc.) it is not so well established. This rule applies to a large number of fruit and vegetables that are known to be a source of vitamins, minerals and/or anthocyanins etc. of undoubted health value," concluded Pecora.
For more information:
pecora.tiziana@gmail.com
